A research team led by Professor Yin-Guo Xiao from the School of Advanced Materials, Peking University Shenzhen Graduate School, has achieved significant progress in the study of Li-rich Mn-based layered oxide cathode materials. The findings are published in Advanced Materials under the title: “Reversible oxygen redox in Li-rich Mn-based cathodes achieved by regulating the local environments of bulk and surface lattice oxygen.” By precisely tailoring the local environments of both bulk and surface lattice oxygen, the team successfully realized highly reversible lattice oxygen redox, substantially enhancing the electrochemical performance of Li-rich Mn-based cathodes. This work provides new insights and an important scientific foundation for the rational design of next-generation high-capacity cathode materials for lithium-ion batteries.
Li-rich Mn-based layered oxides are considered promising high-energy cathode materials due to their dual redox mechanism involving both transition metals and lattice oxygen, delivering ultrahigh specific capacities exceeding 300 mAh g⁻¹. However, the inherently poor reversibility of lattice oxygen redox leads to challenges such as low initial Coulombic efficiency, poor cycling stability, and oxygen release at high voltages, which severely hinder practical applications. To address these key issues, the team proposed a synergistic strategy to regulate the local environments of lattice oxygen in both the bulk and surface regions, achieving highly reversible oxygen redox.
For bulk regulation, the team introduced Li/Mn disorder within the transition metal layers, inducing oriented distortion of MO₆ (M = Li, Ni, Mn) octahedra. This significantly strengthened Mn–O covalent bonding, stabilizing the electronic structure of bulk lattice oxygen while mitigating oxygen framework distortion during electrochemical delithiation. For surface regulation, a mild chemical post-treatment was employed to facilitate Li⁺/H⁺ exchange on the material surface, reconstructing the coordination environment of surface lattice oxygen and effectively suppressing irreversible surface oxygen release.
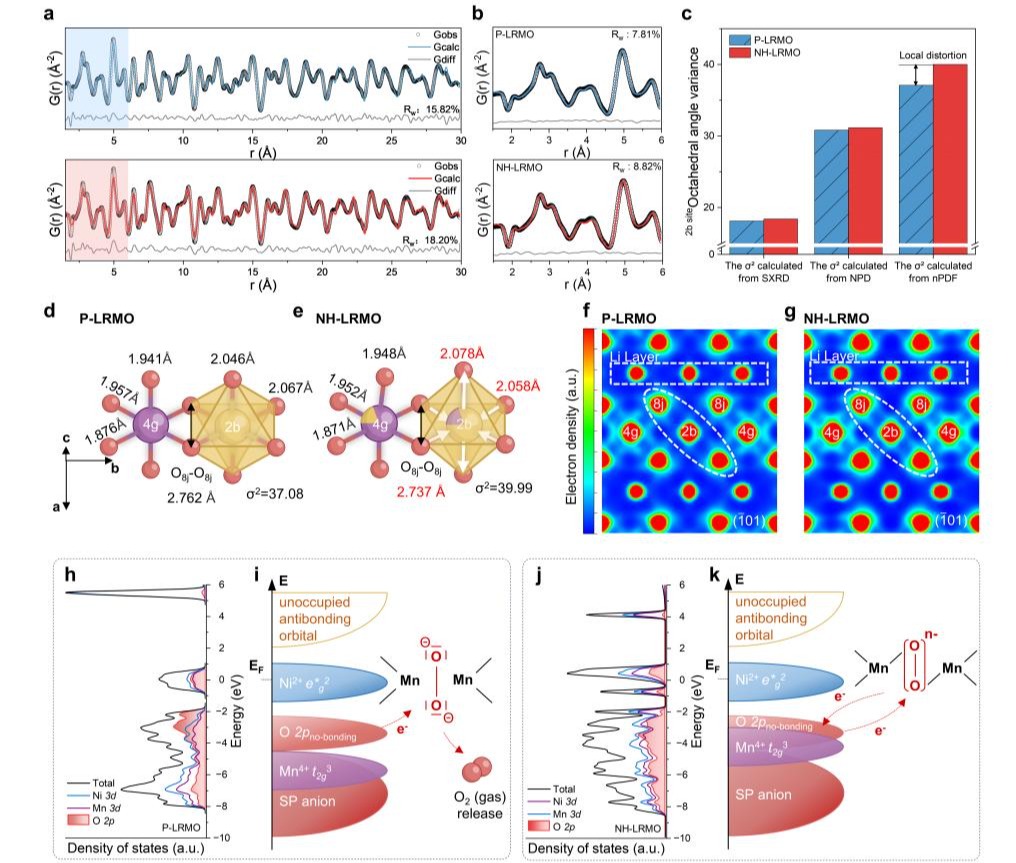
Figure 1. Systematic analysis correlating local structural distortion with electronic structure evolution in the bulk material.
To gain deep insights into the regulation mechanism, the team employed a suite of advanced characterization techniques, including synchrotron X-ray diffraction, neutron total scattering, the maximum entropy method, in situ electrochemical mass spectrometry, and synchrotron soft and hard X-ray absorption spectroscopy. These were complemented by first-principles density functional theory calculations, enabling the establishment of a direct correlation between local structural distortion and electronic structure evolution at the atomic scale. The findings confirmed the enhancement of Mn–O covalent bonding through bulk octahedral distortion and the suppression of lattice oxygen dissociation via surface protonation. In addition, quantitative analysis of Li⁺/H⁺ exchange behavior was achieved using deuterium-labeled high-resolution neutron diffraction, providing direct experimental evidence for the interfacial protonation strategy.
The modified Li-rich Mn-based cathode materials prepared using this synergistic strategy exhibited outstanding electrochemical performance. The initial Coulombic efficiency was significantly increased from 73.88% to 91.72%, with a first-cycle discharge specific capacity of 281.3 mAh g⁻¹. After 500 cycles at a current density of 250 mA g⁻¹, the material retained 95.9% of its capacity, surpassing the performance of previously reported counterparts. Moreover, voltage decay was substantially mitigated, and both the activity and reversibility of lattice oxygen redox were simultaneously enhanced. This study achieves the synergistic stabilization of bulk and surface lattice oxygen in Li-rich Mn-based cathodes, providing a novel strategy to address the critical challenge of improving the reversibility of lattice oxygen redox.
The paper is co-corresponding authored by Associate Professor Yin-Guo Xiao, Dr. Yu-Guang Pu from the School of Advanced Materials, Peking University Shenzhen Graduate School, and Dr. Rui Wang from the University of Cambridge. The co-first authors are master’s student Zi-Qin Jiao and Ph.D. student Tao Zeng from the same school. This research was supported by the National Natural Science Foundation of China, the Guangdong Innovation and Entrepreneurship Research Team Project, and the Open Project of the Songshan Lake National Science Facility in Dongguan, Guangdong. Support was also provided by the Materials Genome Initiative Large-Scale Science Facility Platform, supported by the Shenzhen Municipal Development and Reform Commission.
Link to the paper: https://doi.org/10.1002/adma.202523233
